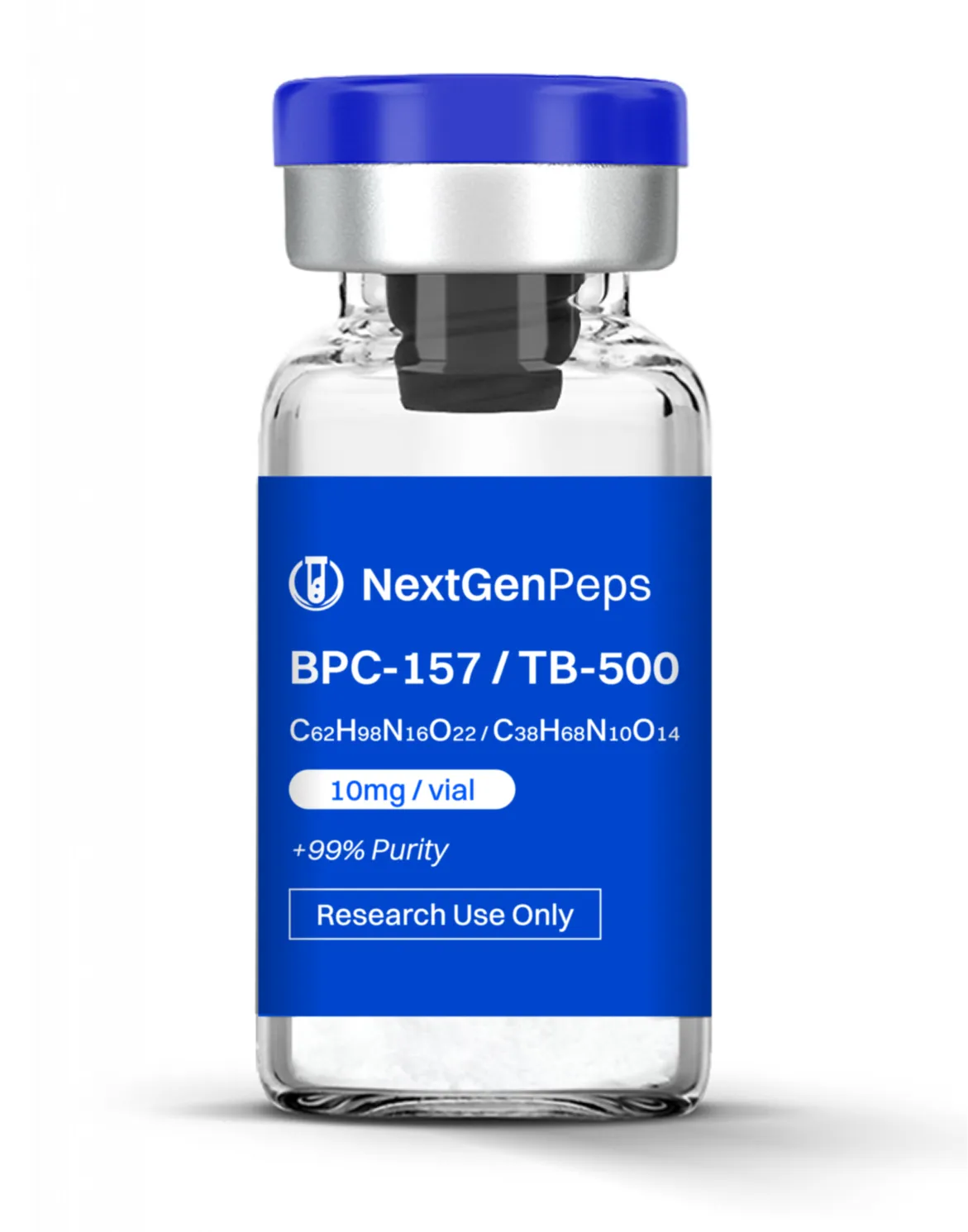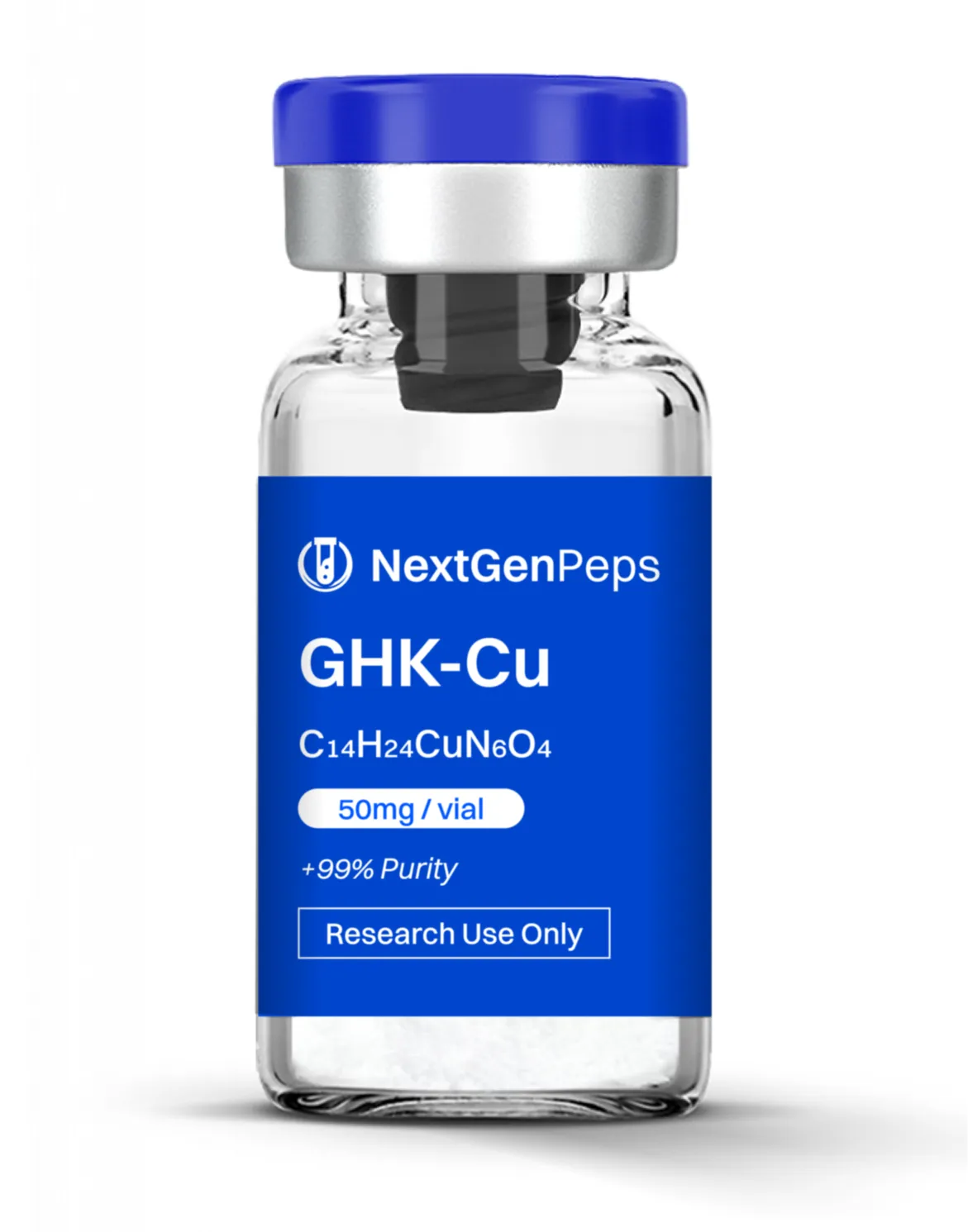
KLOW (Blend)
10mg BPC-157 | 10mg TB-500 | 50mg GHK-CU | 10mg KPV
Out of Stock
Enter your email and we'll notify you when this item is back in stock.
Bulk Options:
Select quantity · Default variant
KLOW (Blend) Research Blend Overview
KLOW is a high-purity, research-grade four‐peptide formulation that combines BPC‐157, TB‐500, KPV, and GHK‐Cu into a single system for advanced investigation of tissue repair, inflammation control, dermal regeneration, and cellular signaling. By integrating peptides that target vascular healing, cytoskeletal dynamics, epithelial integrity, and extracellular matrix (ECM) remodeling, this blend supports multi‐pathway regenerative modeling in controlled laboratory settings.
Molecular Composition and Mechanistic Diversity
Per vial composition (Simple Peptide specification):
GHK‐Cu: 50 mg
BPC‐157: 10 mg
TB‐500: 10 mg
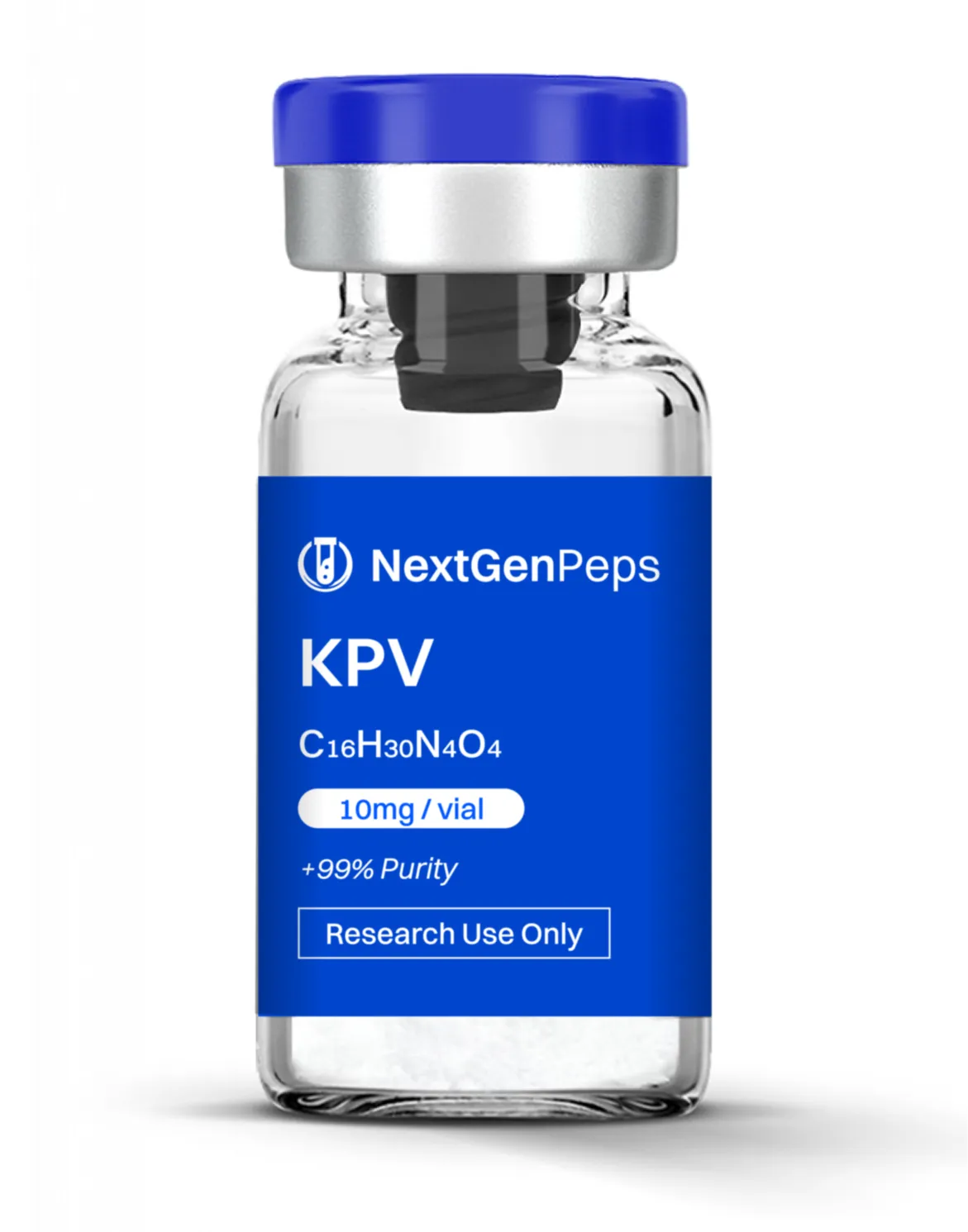
KPV: 10 mg
BPC‐157 (Body Protection Compound‐157)
Supports nitric oxide (NO) signaling, angiogenesis, fibroblast migration, and
cytoprotection in GI, musculoskeletal, and neurovascular models. TB‐500 (Thymosin Beta‐4 Fragment / Timbetasin)
Actin‐binding peptide fragment based on thymosin beta‐4.
Promotes actin polymerization, cell migration, and structural protein expression, aiding soft‐tissue repair, angiogenesis, and reduced fibrosis. KPV (Lys‐Pro‐Val)
C‐terminal tripeptide of alpha‐MSH with strong anti‐inflammatory and immunomodulatory activity.
Investigated for suppression of pro‐inflammatory cytokines, support of gut mucosal integrity, and modulation of innate immune responses.
GHK‐Cu (Copper Tripeptide Complex)
Naturally occurring copper‐binding tripeptide (Gly‐His‐Lys) with potent ECM and gene‐regulatory effects.
Modulates expression of genes involved in collagen synthesis, tissue remodeling, angiogenesis, and antioxidant defense, particularly in skin and connective tissue.
In the KLOW configuration, BPC‐157 and TB‐500 emphasize tissue repair and vascular support, KPV provides anti‐inflammatory and gut‐immune balance, and GHK‐Cu drives collagen and ECM‐centric regeneration, producing a broad regenerative and anti‐inflammatory profile.
Integrated Research Applications
KLOW is formulated for experiments that require parallel study of tissue repair, inflammation, and dermal/ECM remodeling. Representative research areas include:
Muscle, tendon, and ligament recovery: TB‐500 and BPC‐157 support actin remodeling, fibroblast migration, and angiogenesis, while GHK‐Cu contributes to collagen organization and KPV helps moderate inflammatory cascades around injured tissue.
Gut and immune interface: BPC‐157 and KPV are investigated for GI mucosal protection and immune balance, allowing study of epithelial barrier integrity and cytokine modulation under injury or stress.
Skin and aesthetic regeneration: GHK‐Cu dominates dermal regeneration and hair/skin research, with BPC‐157 and TB‐500 augmenting wound closure, vascularization, and reduced scar formation.
Systemic inflammation and recovery: Combined anti‐inflammatory, antioxidant, and pro‐repair actions create a model system for exploring whole‐body recovery after surgery, orthopedic trauma, or intensive training.
Using a single vial with fixed ratios simplifies protocol design and enables evaluation of synergistic versus additive effects across these domains.
Analytical Validation, Formulation, and Storage
Commercial KLOW products are supplied as high-purity lyophilized multi‐peptide vials, typically 80 mg total peptide content per vial (Peptide Sciences listing). Independent testing from another vendor reports closely aligned quantities (≈11.3 mg BPC‐157, 11.1 mg TB‐500, 13.6 mg KPV, and 54.5 mg GHK‐Cu per vial), confirming approximate target ratios.
Key quality attributes:
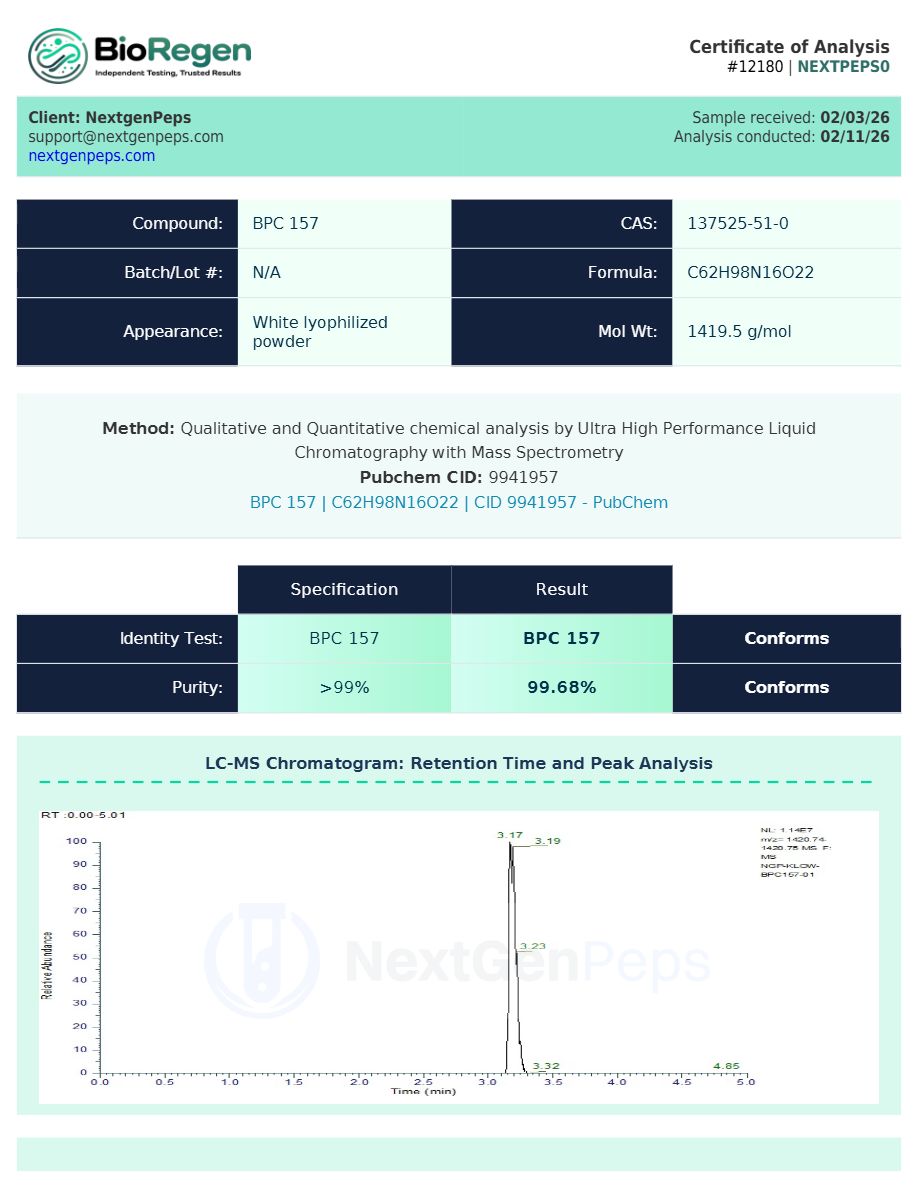
Purity: ≥99% for each component peptide, COA‐verified by HPLC and complementary analytical methods.
Manufacturing: Produced in GMP‐compliant facilities with documented batch testing and endotoxin assessment (e.g., endotoxin “Pass”).
Format: Lyophilized powder in sealed vials, intended for reconstitution with sterile diluent prior to research use.
Storage and handling:
Store lyophilized vials in a cool, dry place away from light to maintain peptide stability.
After reconstitution, refrigerate the solution and, for extended timelines, freeze around −20 °C, minimizing freeze–thaw cycles to preserve integrity.
These practices mirror storage guidance for related blends like GLOW and support consistent behavior over multi‐cycle experiments.
NextGenPeps supplies KLOW exclusively as a research-grade compound for use in controlled laboratory studies. This peptide is not approved for human use and is not intended for therapeutic, diagnostic, or consumable applications.
Frequently Asked Questions
Test Results 80MG
Qualitative and Quantitative chemical analysis for KLOW 80MG by Ultra High Performance Liquid Chromatography with Mass Spectrometry
Similar Products
Reliable and innovative research with our top-grade research materials and novel research compounds tailored to lead the competitive research industry. Choose research quality. Choose NextGenPeps.
401 Wilshire Blvd, Santa Monica, CA 90401
Get 10% Off Your First Order
Sign up now and receive a 10% discount code valid on your next purchase!
NextGenPeps LLC
The statements made within this website have not been evaluated by the US Food and Drug Administration. The materials offered by this company are not intended to diagnose, treat, cure, or prevent any disease. All research materials on this site are for laboratory research and development use only, and are not intended for human use.
NextGenPeps is a chemical supplier. NextGenPeps is not a compounding pharmacy or chemical compounding facility as defined under 503A of the Federal Food, Drug, and Cosmetic act. NextGenPeps is not an outsourcing facility as defined under 503B of the Federal Food, Drug, and Cosmetic Act.
© 2026 NextGenPeps. All rights reserved.